|
Intracranial PFS in patients with advanced NSCLC and brain metastases in the CheckMate 227 trial (Abstract 122MO, ESMO Immuno-Oncology Congress 2021) Neurological treatment-related adverse events (all grade 1/2) were seen in 16% of patients receiving nivolumab plus ipilimumab and 17% of those receiving chemotherapy.įigure. The intracranial objective response rate was the same in both treatment arms (29%), but the duration of response was 45.5 months with nivolumab plus ipilimumab and 32.0 months with chemotherapy. Corresponding 4-year PFS rates were 28% and 7%. Among the patients with brain metastases – which are a common complication of NSCLC – the median intracranial progression-free survival (PFS) was 8.6 months with nivolumab plus ipilimumab versus 8.7 months with chemotherapy (HR 0.80 95% CI 0.50–1.27) (Figure). However, none of the mutations investigated in this analysis were shown to be predictive of response to nivolumab plus ipilimumab.” With the caveat that the patient numbers in some sub-groups are small, he continues, “The results will not have a practice-changing impact, but they do confirm that this treatment option is valid across a variety of tumour mutations.”Ī second sub-group analysis of CheckMate 227 presented at the Congress confirmed that OS benefits of nivolumab plus ipilimumab over chemotherapy were similar in patients with and without treated asymptomatic brain metastases, as previously reported in a post-hoc analysis ( Cancer Res. Professor Benjamin Besse from Institut Gustave Roussy, Villejuif, France, explains: “We already know that STK11 and KEAP1 mutations are associated with a poor prognosis, a finding that was confirmed by this analysis. In the chemotherapy arm, median OS was lower among patients with versus without mutations in the STK11 (11.2 versus 18.5 months) and KEAP1 (8.9 versus 16.7 months) sub-groups, but not in the KRAS sub-group (15.7 versus 17.9 months). Interpretation of the HR of 0.31 among patients with KEAP1 mutations is limited by small patient numbers. Similar survival improvements were seen in patients with and without mutations in KRAS (HR 0.79 and 0.73, respectively) and STK11 (HR 0.78 and 0.75, respectively) and without mutations in KEAP1 (HR 0.80). 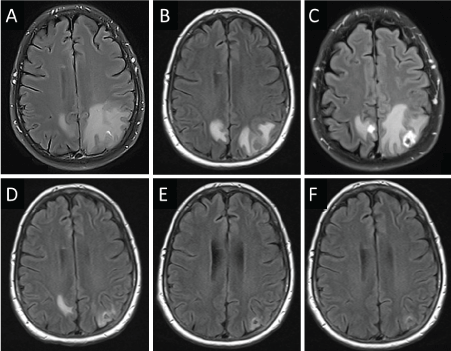
In an exploratory analysis, nivolumab plus ipilimumab improved median OS compared with chemotherapy in 475 patients with non-squamous NSCLC and evaluable mutations, including KRAS (34%), STK11 (16%) and KEAP1 (8%) (20.2 versus 16.3 months hazard ratio 0.75 95% confidence interval 0.61–0.93 4-year OS rates, 32% versus 20%) (Abstract 4O). Two presentations today at the ESMO Immuno-Oncology Congress 2021 report data from sub-group analyses of the trial, with a minimum follow-up of 4 years. 2019 381:2020–2031), with durable efficacy at the 4-year follow-up ( J Thorac Oncol. Maintaining the safety of treatment using immunotherapy targeting two immune checkpoints remains the challenge.The CheckMate 227 part 1 trial previously reported that first-line nivolumab plus ipilimumab prolonged overall survival (OS) versus chemotherapy in patients with stage IV recurrent NSCLC, no known EGFR/ALK alterations and PD-L1 ≥1% (primary endpoint) or <1% ( N Engl J Med. Patients diagnosed with mesothelioma of a non-epithelioid type particularly benefit from two-drug immunotherapy compared to chemotherapy. The results of the CheckMate743 trial for patients with pleural mesothelioma provide a basis for changing the current management algorithm for patients with this diagnosis. A clinical benefit – prolonged overall survival in patients receiving combination therapy – was documented. The CheckMate-9LA study assessed the value of dual immunotherapy combined with platinum-based chemotherapy in the first-line treatment of advanced non-small cell lung cancer. The association of nivolumab with ipilimumab and the interaction of these antibodies on different immune checkpoints have proven effective in solid tumors (melanoma and renal cell carcinoma). 
The value of nivolumab was initially assessed in patients previously treated with systemic therapy. Immune checkpoint inhibitors have significantly changed the treatment of patients with advanced non-small cell lung cancer in recent years.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
